Abstract
Background: Diffuse large B-cell lymphoma (DLBCL) is the most common subtype of malignant lymphoma. Approximately 80% of patients with DLBCL can achieve remission with chemotherapy. However, the prognosis of relapsed and refractory patients is poor. The International Prognostic Index (IPI) is the most widely used prognostic score for non-Hodgkin lymphoma, but it cannot accurately identify a high-risk population for DLBCL. Although gene expression profiles and genomic classifications are other useful tools for predicting outcomes, they are rarely performed in clinical practice. Some antigens, such as CD5 and CD43, have been reported to negatively predict the outcome of DLBCL, while expression of CD10 may have a better prognosis. Most previous studies have been based on immunohistochemistry, in which discordant judgment may occur. Using multicolor flow cytometry, the expression of antigens on lymphoma cells can undoubtedly be documented and surface markers and their combinations may be able to predict prognosis. The present study aimed to develop a new predictive model based on cell surface markers expressed on DLBCL cells.
Methods: Baseline cell surface markers data of the newly diagnosed DLBCL patients enrolled in the present study were analyzed by flow cytometry. The expressions of CD5, CD10, CD19, CD20, CD21, CD22, CD23, CD25, CD38, CD40, CD43, CD44, CD200, and HLA-DR, which are routinely analyzed at our hospital, were obtained for all patients. As the primary endpoint was to determine the 2-year progression-free survival rate (PFS), we applied a decision tree methodology to define which of the abovementioned markers were significant clinical predictors of PFS and established a new cell surface marker-based prognostic score. Based on this analysis, we stratified the patients into good, intermediate, and poor prognosis groups. Survival outcomes for the three groups were estimated using the Kaplan-Meier method and the hazard ratio (HR) and 95% confidence interval (CI) from multivariate Cox regression, considering IPI as a covariate. In addition, the sensitivity and specificity of the new score combined with the IPI score was compared to that of the IPI score alone using the area under the curve (AUC) of receiver operating characteristic (ROC) curves.
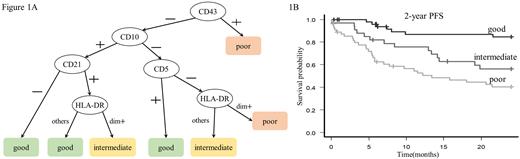
Results: A total of 139 patients with DLBCL were included. The median age at diagnosis was 71 years (range 33-97), and 53% of the patients were male. Regarding IPI scores, 28 patients were in the low-risk group, 28 in the low-intermediate-risk group, 37 in the high-intermediate-risk group, and 46 in the high-risk group. The decision tree method indicated CD5, CD10, CD21, CD43, and HLA-DR as significant predictive variables for 2-year PFS (Figure 1A). The good (51 patients), intermediate (33 patients), and poor (55 patients) prognosis groups identified based on the new score showed a 2-year PFS of 84.5% (95%CI: 70.2-92.3%), 56.3% (95%CI: 37.6-71.4%), and 40.4% (95%CI: 27.0-53.4%), respectively (Figure 1B). The adjusted HR for 2-year PFS was 3.17 (95%CI: 1.27-7.87, P = 0.013) in the intermediate prognosis group and 4.51 (95%CI: 1.97-10.3, P < 0.001) in the poor prognosis group, when compared to the good prognosis group. The ROC analysis showed that adding the new score to the IPI score significantly improved the predictive accuracy of the 2-year PFS from an AUC of 0.688 to 0.767 (P = 0.020).
Conclusion: The new cell surface marker-based risk score for DLBCL patients established here is more useful for predicting the 2-year PFS than the IPI score alone. Further external validation is required to increase the prognostic accuracy of this new score.
Disclosures
Kondo:Asahi Kasei Pharmaceutica,Chugai Pharma, MSD Pharmaceutical, Dainippon Sumitomo Pharma, Otsuka Pharmaceutical: Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal